This post reports findings of a study in Portland, Oregon involving ten volunteers diagnosed with Parkinson’s Disease. Results showed craniosacral therapy relieves symptoms of Parkinson’s. The study design is outlined below along with a summary of findings.
PEP Study Concludes Craniosacral Therapy Relieves Symptoms of Parkinson’s
Results of the Parkinsons Energy Project (PEP) were remarkable given the short time frame – only 4 months -and the limited number of treatments offered. Outcomes were documented quantitatively using standard research instruments, interviews with participants and pre-post videos of motor functions.
I invite you to examine the videos below that show changes in motor functions for several study volunteers that gave us permission to make videos of them public. None of the study participants were on medications so the differences are not due to the effects of prescription medications which can affect how a person feels when they are effective (during the “on” condition) and then they are not (during the “off” condition).
By any-one’s standards, examination of the videos below shows dramatic improvements in motor functions. There is hope of relief from symptoms!
A video of Mary Before the 6 treatments
A Video of Mary After the 6 Treatments
A Video of Steve Before the 6 Treatments
A Video of Steve After the 6 Treatments
A Video of Bob Before the 6 Treatments
A Video of Bob After the 6 Treatments
The encouraging results of the Parkinsons Energy Project (PEP) inspired the development of information, resources and programs that you now find on the Parkinsons Recovery web sites.
Background of the Parkinsons Energy Project (PEP)
Zero Point Healers was asked by a support group in Portland, Oregon in 2005 to conduct a study that evaluated the effects of craniosacral therapy on the symptoms of their members. Members of the support group elected not to take prescription medications for Parkinson’s and were specially interested in exploring treatments and therapies that might potentially offer relief from symptoms. No member of the support group had any prior experience with craniosacral therapy.
Eleven support group members volunteered to participate in the PEP study. Six 30 minute treatments were given every two weeks. One study volunteer dropped out at the mid point of the study because doctors determined his symptoms were caused by Lyme disease.
Treatments were administered by 3 persons who are certified craniosacral therapists and Brennan Healing Science practitioners which is a form of energy work that is complementary to craniosacral work.
Design of the PEP Study
The study was a pre-post design that used quantitative and qualitative measures to assess outcomes.The UPDRS (Unified Parkinson’s Disease Rating Scale) and the Parkinson’s Disease Questionnaire 39 (PDQ-39) were used to assess symptoms quantitatively.
Reliability of both instruments is well documented in the research literature. Instruments were completed by participants before treatments began (the baseline), at the mid point, and after all six treatments had been administered (the post-test).
Video tapes were also taken of 10 motor functions at the beginning of the study and at the end for purposes of comparison. Results of the videos of motor functions were consistent with the quantitative findings.
I will first explain what craniosacral therapy does and why it applies to Parkinson’s I will then summarize the findings of the PEP study.
Craniosacral Therapy
One woman with Parkinson’s who found relief using craniosacral therapy
summarized the nature of the treatments beautifully:
“It is hard to believe anything is really happening, but it works.”
The work is very gentle. The person typically lies down on a comfortable massage table. Craniosacral treatments are not like a massage. The nature of the touch is very, very light and gentle. The person remains fully clothed.
Energy work and craniosacral therapy facilitate the natural ability of the body to unwind and release any tension that is trapped at the cellular level throughout the body. They help the neural system rest and rejuvenate.
The intent of craniosacral therapy is to work with the body so that tension and trauma trapped at the cellular level can be released. Therapists are trained to “follow the body” as they allow tension and stress to be released from the muscles and tissues. The process is best described as one of unwinding tissues that are tightly entangled with too much tension.
Trauma to the body is analogous to a wet wash rag that has been twisted so many times that all the water has been squeezed dry. After the rag is
exposed to the hot sun it dries out and becomes hardened and inflexible. This is what happens to tissues that have been traumatized. Craniosacral therapy softens and helps hydrate the tissues so that they can be properly
nourished.
Why Craniosacral Therapy Relieves Symptoms of Parkinson’s
Stress is a key factor that aggravates symptoms. When stress can be relieved, symptoms get better.
Person after person in my interviews tell me the same thing: There is a direct correlation between stress and their symptoms.
Symptoms flare up when confronted with stressful situations. When stress is no longer a compelling influence symptoms become less troublesome.
Stress creates inflammation throughout the body, the basis for all chronic
diseases. Reduction in inflammation is why symptoms improve. There is a three step sequence involved:
(1) Craniosacral therapy releases stress.
(2) Inflammation is reduced.
(3) Symptoms are relieved.
Craniosacral therapy is only one way to help relieve stress. There are also
other excellent methods that help to relieve stress, but we zeroed in on craniosacral work as the intervention of choice in the PEP study.
Trauma and Parkinson’s
Life experiences can be so traumatic that they have a long lasting effect on the body. The source of trauma varies widely, but can involve the early and untimely death of a parent, service during wartime, physical or sexual abuse, automobile accidents, head injuries – you name it.
I was discussing trauma this week to a Parkinson’s support group when one woman
spoke up. .
“I know an example of this. I have a friend whose son committed suicide. His symptoms flared up two months later.”
Regardless of the source, trauma makes the tissues throughout the body freeze up. Tissues in many people with Parkinsons feel much like concrete after it has hardened.
Many adults (myself included) have held the belief that if we are able to function in the world as adults by educating ourselves, getting good jobs, and making a contribution n our respective fields, we have overcome any and all traumas we experienced as a child.
I personally convinced myself for 50 years that I could “will” traumas from my own childhood to vanish into thin air. Speaking from personal experience, I must confess this plan was a big flop.
What I now know to be true is that traumas from childhood (and traumas we experience as adults) have a permanence about them. Traumas reside inside our cells as we age. They get stuck in our tissues and settle in for the long haul. This is why our issues are in our tissues.
When a threshold level of trauma and stress is reached the body becomes less and less able to tolerate it. The good news is that it takes time for the hormones in the body to become seriously imbalanced. It should thus come as little surprise that it also takes time for hormones to come back into balance.
Findings of the PEP Study Reveal Craniosacral Therapy Relieves Symptoms of Parkinson’s
Findings of our study showed that craniosacral work helps to relieve Parkinson’s symptoms. Quality of life among study volunteers also improved. Study volunteers were able to do things that they had previously been unable to do. Here are some examples:
- Cook Thanksgiving dinner
- Take weekly nature hikes
- Go out on a date
- Travel
- Be assertive
- Take off a coat without assistance
- Stand up to brush teeth
- Attend business meetings
- Swing arms freely
- Blink eyes
- Hang out with friends
- Drive without anxiety
- Handle stress better
We observed other positive changes from one treatment session to the next including:
- More flexibility getting on and off the treatment table
- Better posture
- Greater ease walking
- Enhanced mental clarity and expressiveness
- Greater flexibility
- More smiling, better skin color and overall vibrancy
- More vitality and youthfulness
Craniosacral therapy clearly had a positive impact on the overall health and well being of study volunteers.
Unified Parkinsons Disease Rating Scale (UPDRS).
The quantitative research instruments confirmed reports from study participants, footage from videos and our own observations. One standard instrument used in Parkinson’s research to evaluate the progress of Parkinsons is the Unified Parkinsons Disease Rating Scale (UPDRS).
The UPDRS was administered before the study began and at the conclusion of the study six months later. The average baseline score for the PEP study group was 40.3. The average score dropped to 38.7 at the conclusion of the study.
The higher the score, the worse the condition. That is to say, study participants got better on average. Some experienced significant improvements. Further analysis of the data showed that study volunteers improved on all three sub-scales of the UPDRS (Activities of Daily Living, Mental, and Motor).
Most people believe that the symptoms of people with Parkinson’s are “destined” to progressively “deteriorate” over time. Results of the PEP study refute this widely held belief and show it to be false.
The improvement was remarkable in that the intervention was modest, involving only 6 treatments lasting 20-30 minutes each.
Changes in Symptoms.
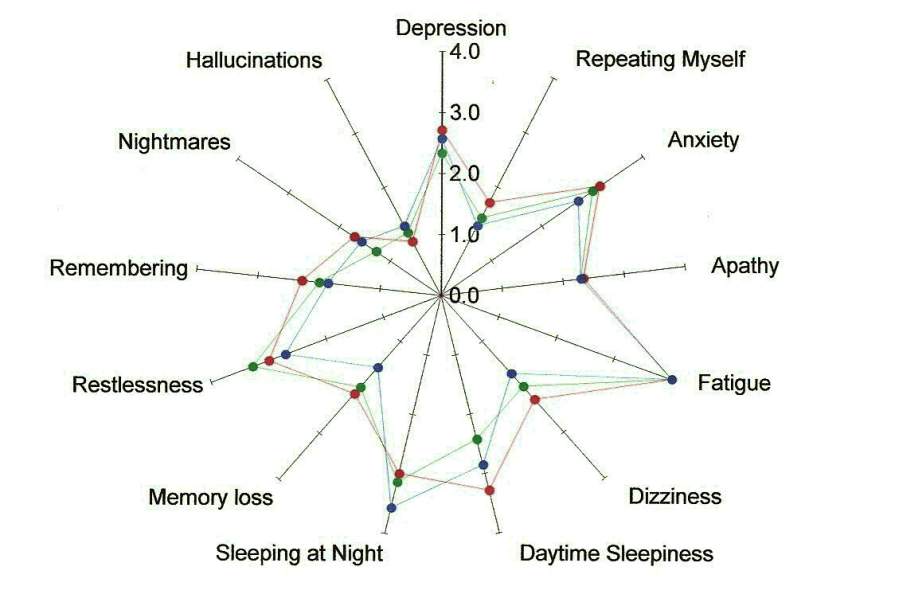
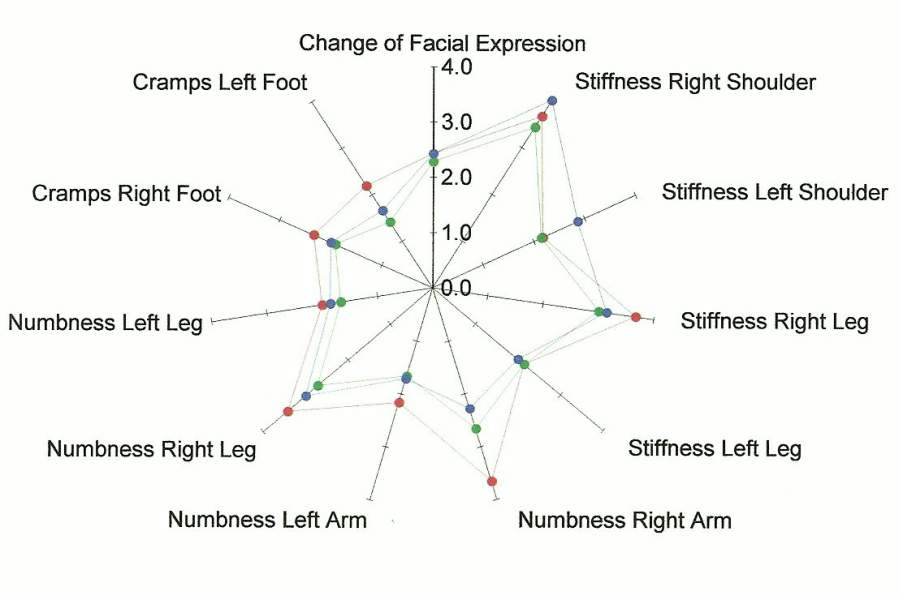
We also asked Study Volunteers to report on 62 symptoms before the study began, at midterm and at post test. Analysis across the three time periods revealed there was improvement or no change in 75% of the symptoms we tracked.
In the figures below, the center point is the place where the person is symptom free. The farther out from the center of each spoke of the wheel, the worse the person is feeling.
The points marked in red reflect the averaged baseline symptoms for study volunteers. The green points are the averaged mid term report of symptoms taken after three treatments. The blue points are the averaged post-test report of symptoms taken after 6 treatments.
For many of the symptoms, the color of the dot that is furthest from the center (reflecting a worse condition) is red. Note that the plot point in blue that represents symptoms at the end of the study are usually closer to the center than the baseline in red. This indicates an improvement in symptoms.
Parkinsons Disease Questionnaire 39.
We also administered a third instrument – he Parkinson’s Disease Questionnaire 39. Among data reported on this questionnaire, the sub-scales analyzed from the data reported on this questionnaire, improvement was seen in two categories, no change was seen in four categories and Study Volunteers were slightly worse in two categories. Again, these findings refute the conventional expectation that the condition of all persons with Parkinson’s is destined to deteriorate.
Summary: Craniosacral Therapy Relieves Symptoms of Parkinson’s
All sources of evidence including quantitative data and qualitative observations revealed consistent findings. It is possible to celebrate relief from the symptoms of Parkinson’s. Craniosacral therapy is a viable option which can offer symptom relief.
therapy is a viable option that offers symptom relief. When trauma embedded at the cellular level is released, symptoms improve.
Robert Rodgers, Ph.D.
Founder 2004
Parkinsons Recovery®
© 2024 Parkinsons Recovery